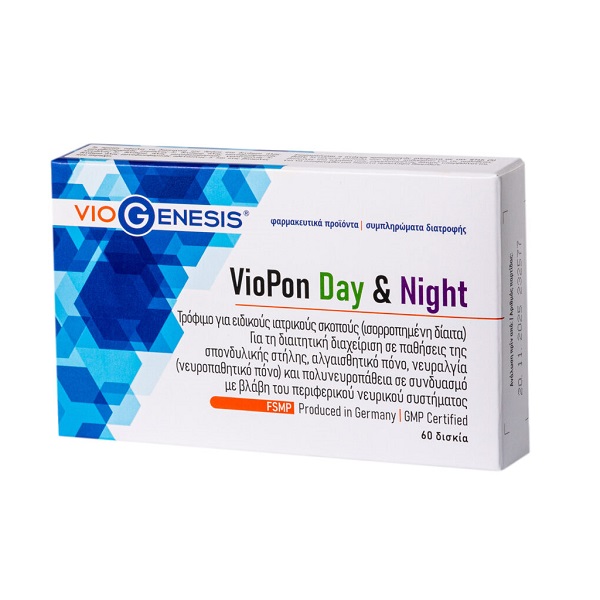
VioGenesis VioPon Day & Night 60 tabs
31,02 €
Food for special medical purposes
1 in stock
Food for special medical purposes (balanced diet). For the dietary management of spinal syndromes, algesic syndromes, neuralgia (nerve pain) and polyneuropathy associated with damage to the peripheral nervous system.
Contains: 60 tablets (30 tablets Day + 30 tablets Night). Folded box containing one blister card of 30 tablets of Day and one blister card of 30 tablets of Night.
Under European Directive (EU) 609/2013 & (EU) 128/2016 FSMP [Food for Special Medical Purposes] are defined as dietary products [balanced diet], which are intended for the dietary management specific for a disease, disorder or medical condition. Food for special medical purposes are based on clinical trials of the active ingredients they contain and are used under medical supervision.
In particular, according to the European directive, they are intended for patients with other medically-determined nutrient requirements, whose dietary management cannot be achieved by modification of the normal diet alone.
Food for special medical purposes are more practical and safer than regular foods.
More information on Directive (EU) 609/2013 & (EU) 128/2016 can be found on the official website of the European Parliament and of the Council www.eur-lex.europa.eu.
VioGenesis Food for special medical purposes are nutritionally incomplete food with a nutrient-adapted formulation specific for a disease, disorder or medical condition which is not suitable to be used as the sole source of nourishment.
How To Use
Recommended dosage: Take one tablet Day in the morning and one tablet Night before sleeping with water daily.
Νutritional supplements do not replace a balanced diet. Consult your doctor or pharmacist.
| Active Ingredients Day | per 1 tablet Day | % NRV* |
| Uridine 5′ – monophosphate disodium salt | 50 mg | ** |
| Cytidine 5′ – monophosphate disodium salt | 10 mg | ** |
| Guanosine 5′ – monophosphate disodium salt | 2.50 mg | ** |
| Adenosine 5′ – monophosphate disodium salt | 2.50 mg | ** |
| Vitamin B12 | 187.5 μg | 7500 |
| Vitamin B6 | 1.4 mg | 100 |
| Folic Acid | 200 μg | 100 |
* % Nutrient Reference Value (NRV) according to the regulation EU 1169/2011
** No Reference Value available
| Active Ingredients Night | per 1 tablet Night | % NRV* |
| Uridine 5′ – monophosphate disodium salt | 50 mg | ** |
| Cytidine 5′ – monophosphate disodium salt | 10 mg | ** |
| Guanosine 5′ – monophosphate disodium salt | 2.50 mg | ** |
| Adenosine 5′ – monophosphate disodium salt | 2.50 mg | ** |
| Vitamin B12 | 187.5 μg | 7500 |
| Vitamin B6 | 1.4 mg | 100 |
| Melatonin | 1 mg | ** |
| Folic Acid | 200 μg | 100 |
* % Nutrient Reference Value (NRV) according to the regulation EU 1169/2011
** No Reference Value available
Aditional Information
| Weight | 0,100 kg |
|---|---|
| Age group | |
| Brand |

 Ελληνικά
Ελληνικά